ESBL RESISTANCE DETECTION

NG-TEST® CTX-M Multi
Results within 15 minutes
NG-TEST® CTX-M Multi is a qualitative lateral flow immunoassay for the rapid detection of the five major groups in the CTX-M-type enzymes of extended-spectrum β-lactamases (ESBLs) produced by Enterobacterales, from a bacterial colony. The Rapid Tests detects enzymes belonging to CTX-M Groups 1, 2, 8, 9 and 25 including their most clinically relevant variants in the same cassette in less than 15 minutes from a bacterial colony .
| Product Name | Targets | Description | Technology | Reference |
|---|---|---|---|---|
| NG-TEST® CTX-M Multi | CTX-M Groups 1, 2, 8, 9 & 25 | 20 cassettes, buffer, 20 microtubes, 20 pipettes | Lateral flow | NGB-CTM-S23-016 |
Over the past several years, numerous scientific studies have been conducted and peer-reviewed supporting the exciting capabilities of our rapid tests.
Please take a moment to complete the following information and you will receive the publications via email.
Certain uses of devices included in these publications may not have been approved or cleared by the U.S. Food and Drug Administration or other regulatory bodies.
See product labelling for instructions for use, warnings, and limitations specific to each product and region.
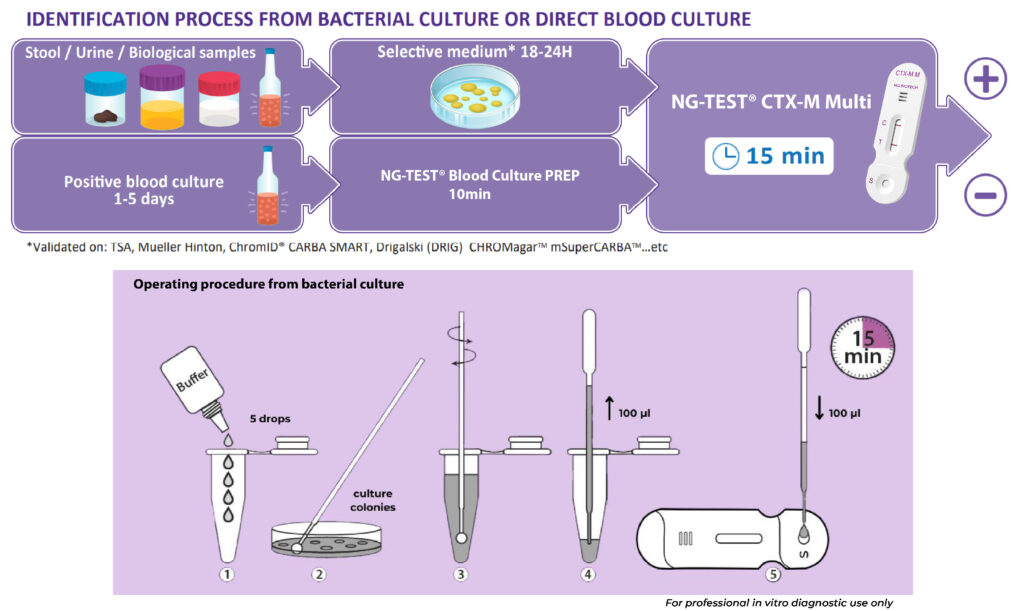
WORKFLOW
NG-TEST® CTX-M Multi is a rapid in vitro diagnostic assay, for professional use only. It is a visual multiplex immunochromatographic (lateral flow) qualitative assay for the detection of the five CTX-M groups 1, 2, 8, 9 and 25 from extended-spectrum β-lactamases (ESBLs) pure bacterial colonies of Enterobacterales (including Escherichia coli and Klebsiella pneumoniae) in healthcare settings.
The use of NG-TEST® CTX-M Multi in the laboratory provides information to inform appropriate antimicrobial therapeutic choices and support prompt infection control decision making and interventions.
The NG-TEST® CTX-M Multi can be easily implemented in any laboratory setting with minimal training.
It requires minimal hands-on time, the preparation protocol is simple and no instrumentation is required. NG-TEST® CTX-M Multi can also be performed directly from Positive Blood Culture samples, which can support treatment decisions about 1-2 days faster than current methods allow today.
RELATED PRODUCTS
Due to the increase in the percentage of infections caused by Multi-Drug Resistant (MDR) bacteria, more information is needed about the expressed mechanisms of resistance to help guide the optimal antibiotic treatment and to manage infected patients appropriately.
At NG Biotech we continuously develop complementary products to provide additional clinically relevant information to adequately treat patients and/or implement containment measures.
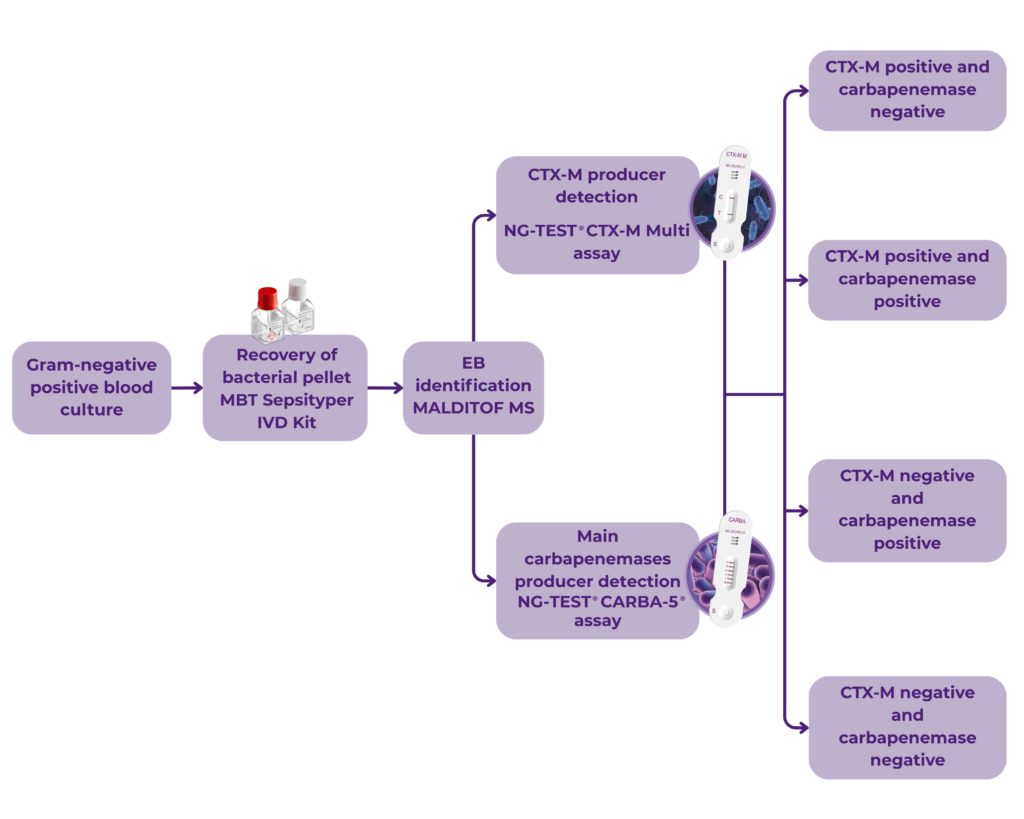
PROCESS
A recent study by Boattini et al. 2021 presented a fast-track diagnostics workflow for the detection of ESBL CTX-M and Carbapenemase producers directly from Enterobacterales Positive Blood Cultures followed by the implementation of the algorithm in their laboratory regular workflow.
SPECIFICATIONS
Sensitivity: 100% Specificity: 100%
NG-TEST® CTX-M detects the following variants:
- Group 1: CTX-M-1, -3, -10, -15, -32, -37, -55, -57, -71, -82, -101, -182
- Group 2: CTX-M-2
- Group 8: CTX-M-8
- Group 9: CTX-M-9, -13, -14, -17, -18, -19, -24, -27, -38, -65, -93
- Group 25: CTX-M-94, -100