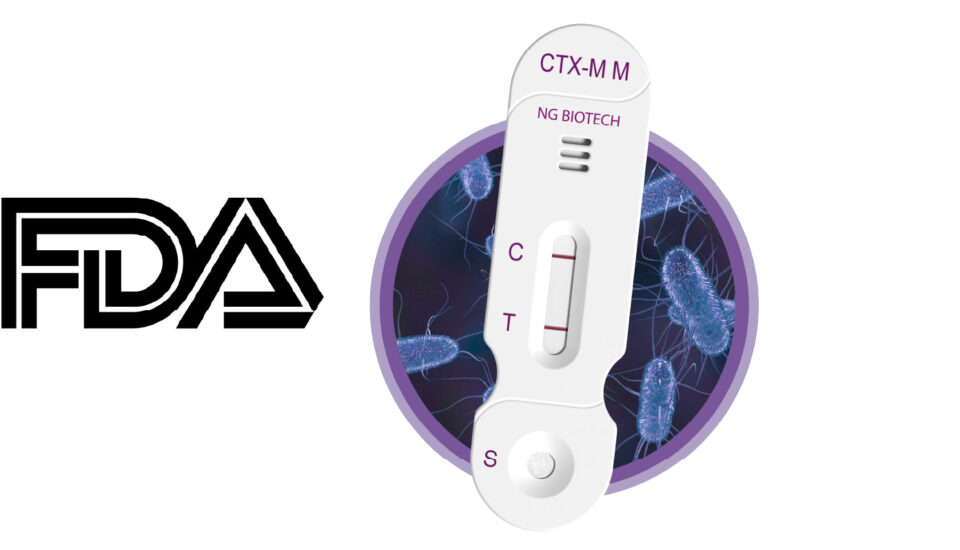
FDA Breakthrough Device Designation – 2026 FEBRUARY 18
FDA Grants Breakthrough Device Designation to Two NG Biotech Rapid Tests Targeting the World’s Most Dangerous Drug-Resistant Pathogens A designation recognizing innovation against WHO-critical fungal and bacterial threats Santa Maria, CA, USA / Guipry, France, February 18, 2026; In a significant recognition of innovation in the fight against Antimicrobial Resistance (AMR), NG Biotech, in partnership […]